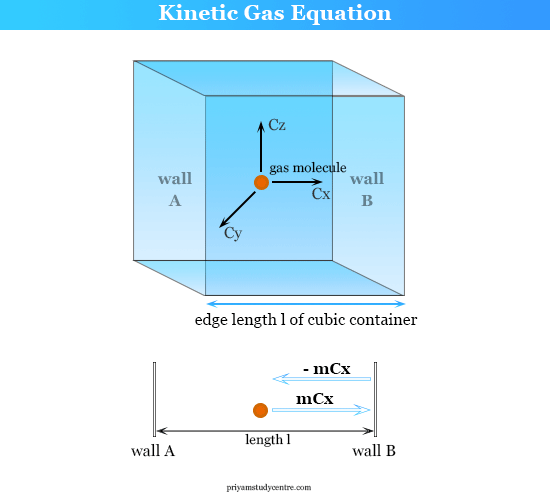
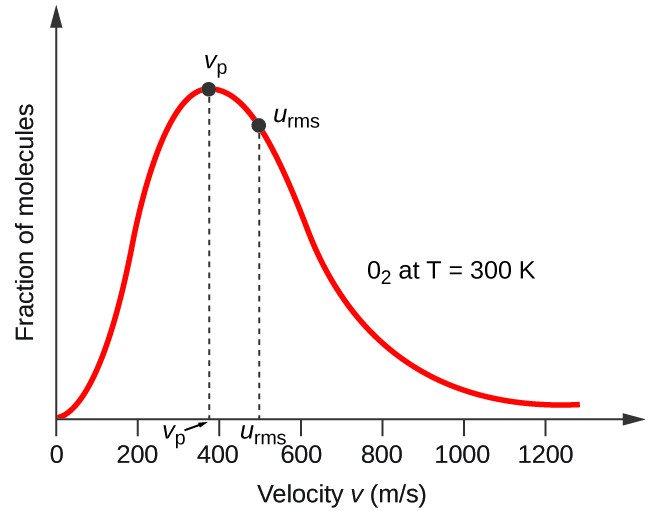
Calculate root mean square speed, most probable speed and average speed of : O2 if 6.431 g of it occupies 5 litres at 750 mm.

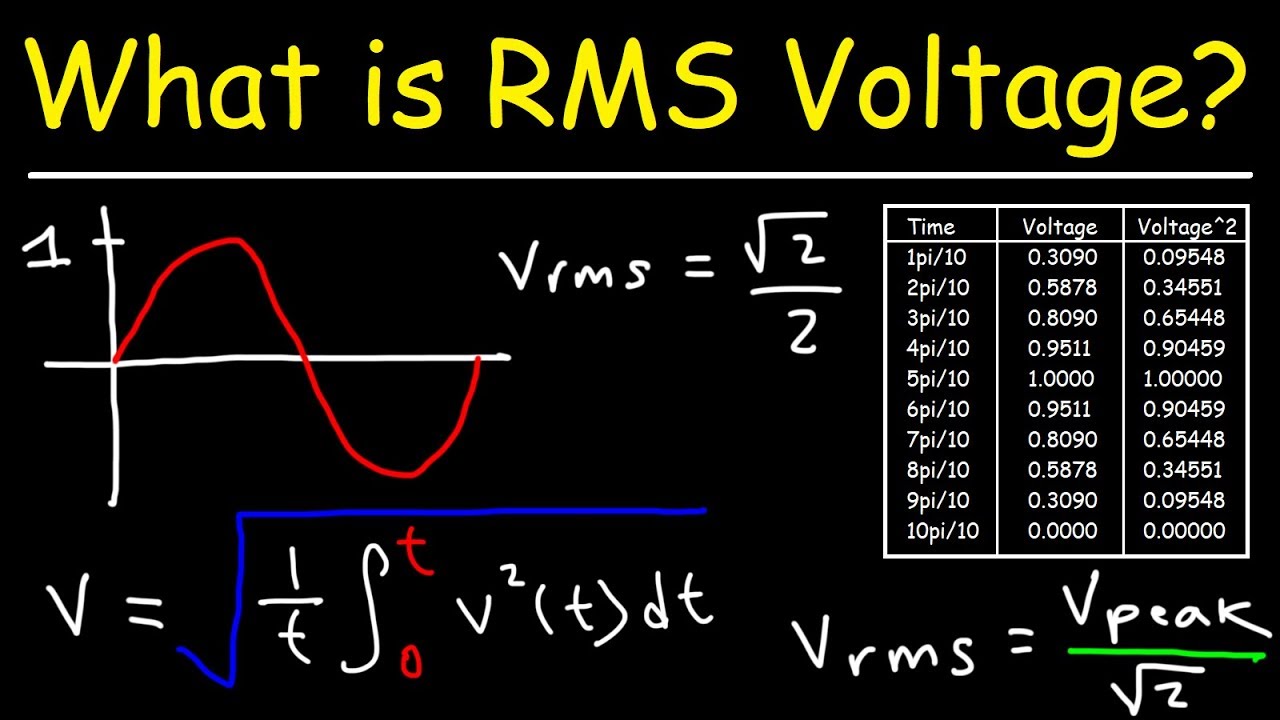
How to Calculate the Root Mean Square Speed of Molecules in Gas at a Certain Temperature | Physics | Study.com

RMS Value, Average Value, Peak Value, Peak Factor, Form Factor in AC | Rms, Engineering notes, Electrical circuit diagram
At what temperature is the root mean square speed of nitrogen molecules equal to the root mean square of hydrogen molecules at 20 degree Celsius? - Quora






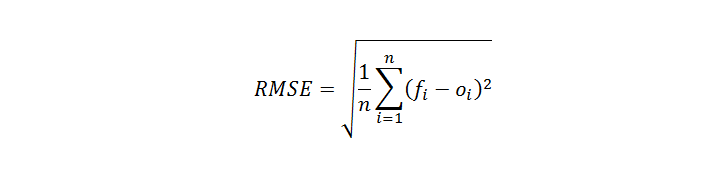



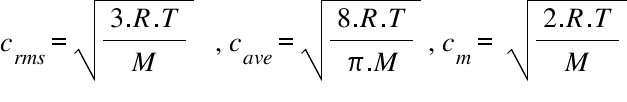

:max_bytes(150000):strip_icc()/ChalkboardCalculations-58b1c7c43df78cdcd8148e15.jpg)